Protein in vaccine8/10/2023 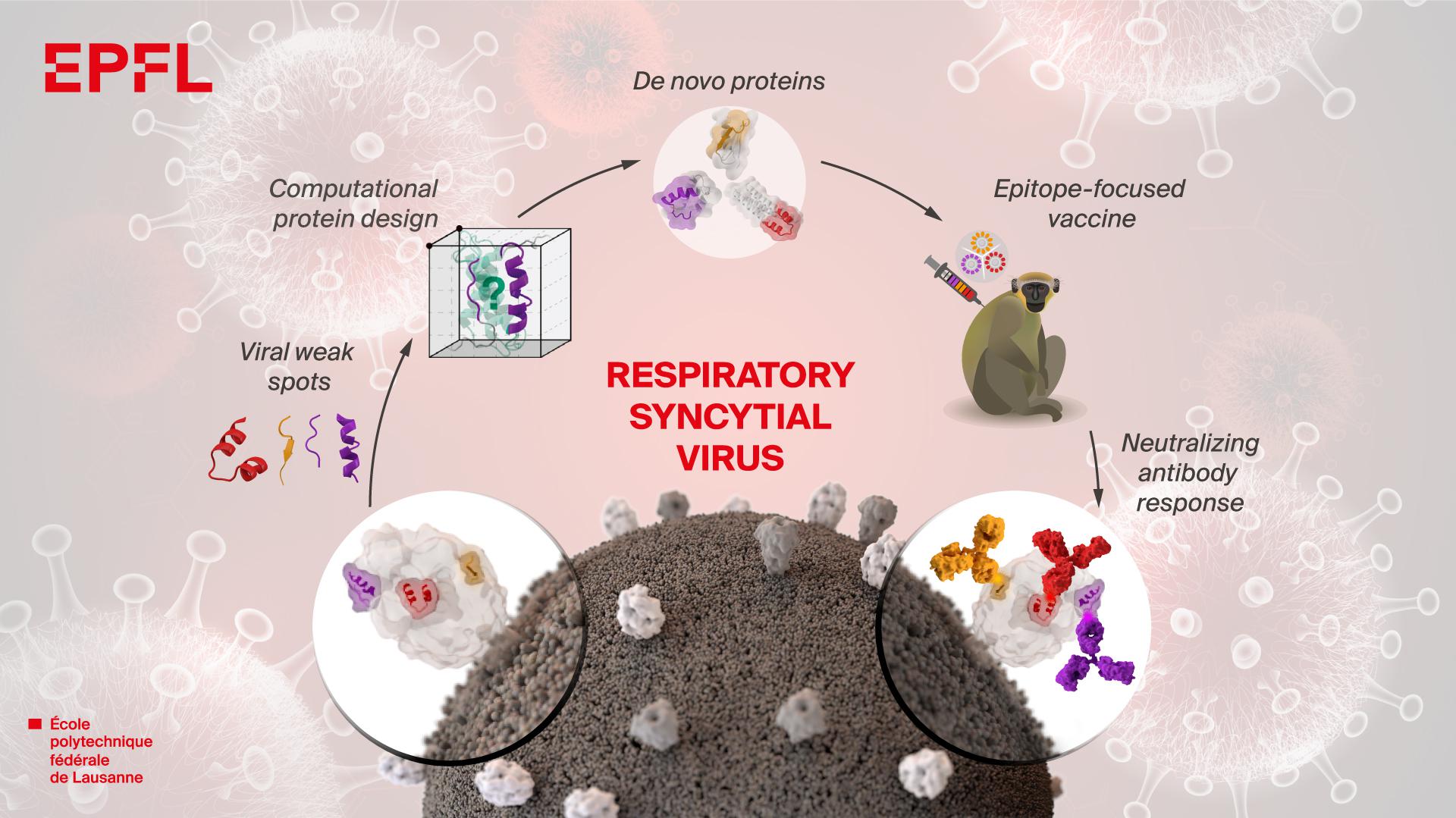
Another widely used and successful VL drug Miltefosine tends to have teratogenic effects in animals. Application of Amphotericin B provides a cure rate of more than 98 percent but causes fever, chills, thrombophlebitis, and in serious cases nephrotoxicity. Pentavalent Antimonials which are widely used drugs for VL is being proven less effective due to drug resistance mainly in India. Currently, anti-leishmanial therapies, based on chemotherapeutics are available but drug toxicity and numerous side effects set a huge drawback. More than 50,000 annual deaths due to leishmaniasis have been reported by The Global Burden of Disease Study (GBD) in 2010. In these countries, leishmaniasis is a significant health issue and sets a huge impact on socio-economic conditions. 90% of worldwide visceral leishmaniasis cases have been reported in Ethiopia, India, Bangladesh, South Sudan, and Sudan, Brazil, Kenya. The disease is endemic in more than 60 countries, with an estimated 50,000–90,000 new cases of visceral leishmaniasis occurring each year worldwide. Visceral Leishmaniasis (VL) is the deadliest form of leishmaniasis which is caused by Leishmania donovani in South Asia (India, Bangladesh, Nepal) and East Africa (Sudan, Ethiopia, Somalia, and Kenya) and by Leishmania infantum in the Middle East, Mediterranean, Brazil, Pakistan and Iran. Sandflies mainly of the genus, Phlebotomus and Lutzomyia are known to spread this disease. The genus Leishmania belongs to a family of Trypanosomatidae of the order kinetoplastida. Leishmaniasis is a vector-borne chronic inflammatory disease, caused by the protozoan parasite of the Leishmania genus, transmitted to humans by sandflies. Further wet lab synthesis of the vaccine and in vivo works has to be undertaken in animal model to confirm vaccine potency. The above bioinformatics data support that the construct may act as a potential vaccine. In-silico codon optimization and cloning of the vaccine nucleic acid sequence have also been achieved in the pET28a vector. 
In-silico immune simulation studies confirms the vaccine to raise a pro-inflammatory response with the proliferation of activated T and B cells. 
Population coverage analysis shows the vaccine can be effective on the majority of the world population. The predicted tertiary structure of the vaccine has been found to be valid and docking studies reveal the construct shows a high affinity towards the TLR-4 receptor. The vaccine construct is generated by joining the epitopes with specific linkers. 
The various physicochemical characteristics of the vaccine have been explored and the tertiary structure of the chimeric construct is predicted to perform docking studies and molecular dynamics simulations. We have chosen four antigenic proteins of Leishmania donovani and identified their T-cell and B-cell epitopes, utilizing those for in-silico chimeric vaccine designing. With the advancement of bioinformatics and its application in immunology, it is now more convenient to design multi-epitope vaccines comprising predicted immuno-dominant epitopes of multiple antigenic proteins. Conventional vaccine synthesis techniques are often very laborious and challenging. VL falls under the category of neglected tropical diseases with growing drug resistance and lacking a licensed vaccine. Visceral Leishmaniasis (VL) is a fatal vector-borne parasitic disorder occurring mainly in tropical and subtropical regions.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
